Reduced exposure to HPHCs in smokers who switch from cigarettes to the Tobacco Heating System
How were PMI’s reduced exposure studies conducted?
Our two 5-day reduced exposure studies were conducted in Japan (clinicaltrials.gov: NCT01970982) and Poland (clinicaltrials.gov: NCT01959932, and another related publication). Our two 90-day reduced exposure studies used the menthol version of THS and were conducted in Japan (clinicaltrials.gov: NCT01970995) and in the U.S. (clinicaltrials.gov: NCT01989156). These were all 3-arm studies involving 160 participants, with approximately 80 participants randomized to switch to THS for the duration of the study, and approximately 40 participants each assigned to continue smoking their usual brand of cigarettes, or to quit smoking for the duration of the study. THS was provided for those assigned to the THS arm, and those assigned to the cigarette arm continued to purchase their usual brand of cigarettes. The smoking abstinence arm participants’ abstinent status was verified by exhaled carbon monoxide levels.
All studies began similarly, with participants spending five days confined to the study site. This confinement period allowed for strict control over the participants’ use of their designated product by preventing the participants from using products they were not assigned. However, because the confinement period was only five days, this reduced the chance that a reduction in biomarkers with longer half-lives would be detected, such as total NNAL, for which the half-life ranges between 10 and 18 days. NNAL is itself a tobacco-specific nitrosamine and is a biomarker for NNK which is also a tobacco-specific nitrosamine.
After the 5 days in confinement, the 3-month studies continued with the participants returning home with instructions to continue to follow the instructions for their study group as they had already done for 5 days at the study site: continue to smoke their own brands of cigarettes, continue using THS, or continue to abstain from cigarettes. The participants returned for measurements each month and stayed overnight at the study site on days 30, 60, and 90. This longer study period made it more likely that a reduction in biomarkers for constituents with longer half-lives, like NNAL, could be detected. Further, the exposure reduction effects for THS could be examined in real-life conditions. The participants self-reported their product use during this ambulatory period outside of the clinical setting.
What are biomarkers of exposure?
A biomarker of exposure is something that is measured in the body that indicates exposure to some stimulus. To give an example from tobacco research: the presence of a biomarker of exposure for nicotine, cotinine, indicates that the person was exposed to nicotine. This connection comes from the fact that nicotine gets metabolized, or converted, to cotinine in the body. And cotinine is one of several metabolites of nicotine, which is an example of why multiple biomarkers of exposure can be used for the same chemical, even within the same study.
Biomarkers of exposure are typically found in the blood, saliva, or urine, and they need to be specific to the chemical being studied. It may be a metabolite, as described above, or the result of some other kind of chemical reaction inside the body. For example, carbon monoxide exposure can be measured based on the presence of carboxyhemoglobin (COHb), which is carbon monoxide that is bound to hemoglobin. Hemoglobin is a protein that carries oxygen from the lungs to other parts of the body and carries carbon dioxide back to the lungs. But it binds more tightly to carbon monoxide, making COHb a useful biomarker of exposure to carbon monoxide. Nicotine and carbon monoxide were some of the constituents that were examined in these reduced exposure studies.
What biomarkers were measured in the reduced exposure studies?
In each of the studies, the same 15 harmful and potentially harmful constituents (HPHCs) were assessed using specific biomarkers of exposure in each case. In addition to these 15, the studies also tested for biomarkers of exposure to nicotine: nicotine and cotinine in the blood plasma, and nicotine equivalents measured in 24-hour urine samples. These HPHCs were selected for these studies according to specific criteria.
Read the criteria for biomarker of exposure selection in these reduced exposure studies:
- HPHCs recommended for mandated lowering in cigarette smoke as defined by the World Health Organization (WHO Study Group et al., 2008) and the draft guidance of the U.S. Food and Drug Administration (FDA) Center for Tobacco Products (CTP) on “Reporting Harmful and Potentially Harmful Constituents in Tobacco Products and Tobacco Smoke” (U. S. FDA , 2012).
- The HPHC is specific to cigarette smoke with other sources being minor or nonexistent.
- The biomarker of exposure to a HPHC is easily detectable using validated, reliable, reproducible, and precise analytical methods.
- The HPHC reflects a specific toxic exposure or is a reliable surrogate of exposure to HPHCs.
- The list of HPHCs includes HPHCs from both the gas and particulate phases.
- The list of HPHCs includes a broad variety of chemical classes and organ toxicity classes as defined by the U.S. FDA (carcinogen, cardiovascular toxicant, respiratory toxicant, reproductive and development toxicant, addiction potential) (U.S. FDA , 2012).
- The list of HPHCs includes HPHCs formed at different temperature levels.
- The HPHCs selected for measurement vary in their elimination half-life times, ranging from a few hours up to more than 2 weeks.
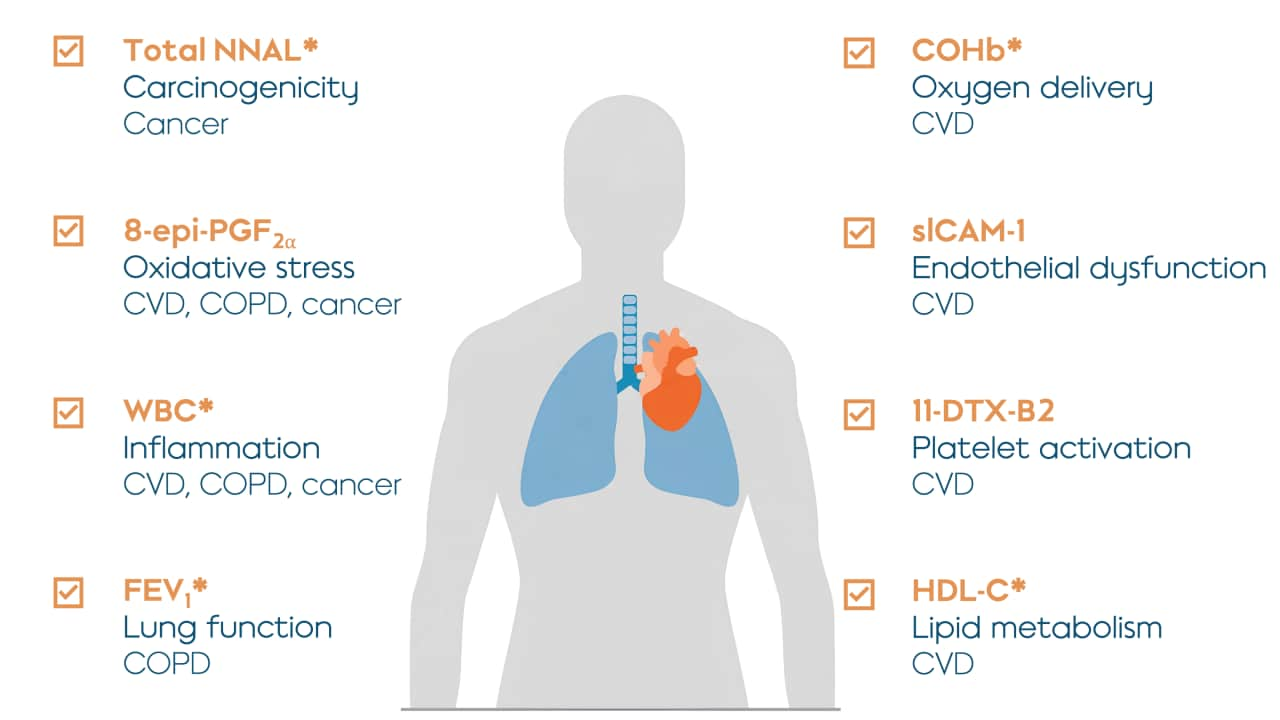
The list below shows the 15 HPHCs in these studies, their biomarkers of exposure, and the related health concern based on information available on the U.S. FDA website.
The studies also measured levels of S-benzylmercapturic acid or S-BMA, which is a biomarker of exposure to toluene, with only subtle differences observed between smokers and nonsmokers.
In addition to these biomarkers, several subjective effects were also measured as part of these studies. More study details and results can be found at the relevant publication links:
What are the results of the reduced exposure studies?
In short, the levels of the 15 biomarkers of exposure to HPHCs in the study participants were reduced among those who switched to THS from cigarettes, and the levels of those biomarkers stayed low during the length of the studies. The levels of biomarkers of exposure for participants who switched to THS were comparable to the levels of those who quit smoking for the duration of the study. For both the THS group and the smoking abstinence group, the levels of biomarkers were significantly lower than those observed in participants who continued smoking for the duration of the study.
Switching from cigarettes to THS for 5 days resulted in the reduction of 15 biomarkers of exposure in the THS group vs the cigarette group ranging from 56% to 96%. Those values approached the exposure reduction observed in the overall smoking abstinence group. Similar observations were made for the 90-day studies, where the reductions observed at day 5 were maintained through the end of the 3-month studies.
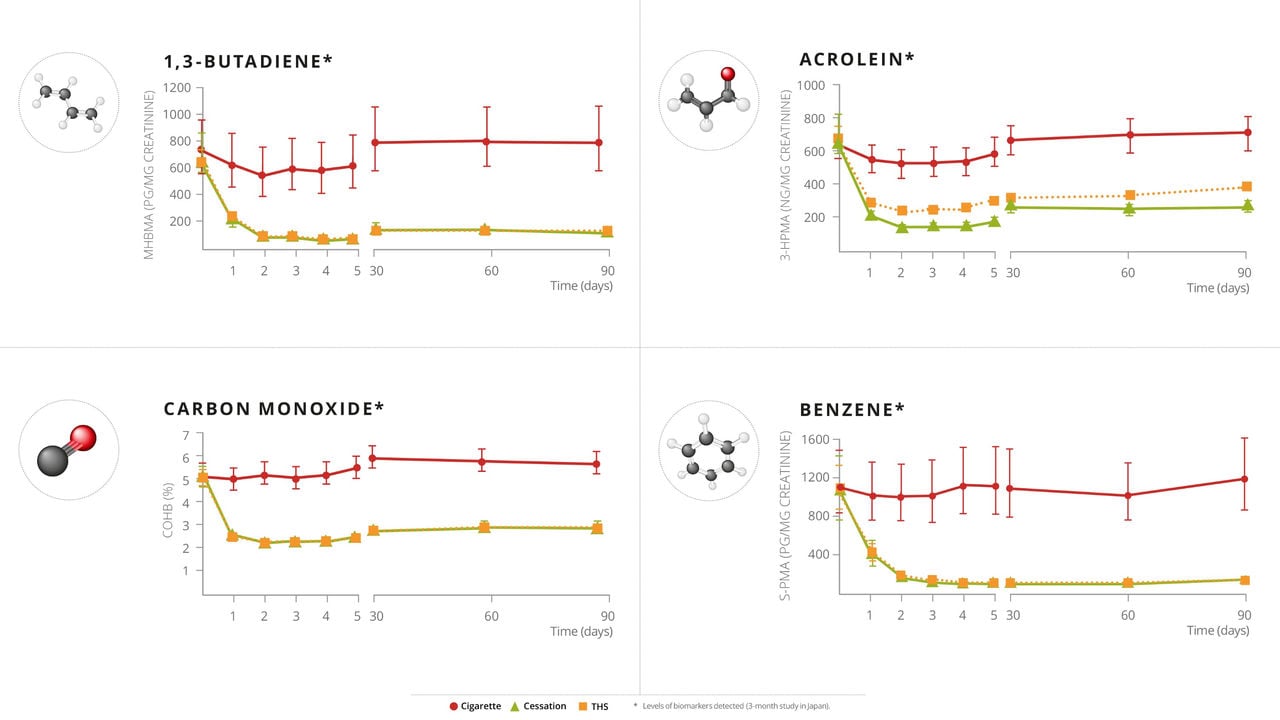
Graphs showing the levels of four biomarkers of exposure measured over the course of the 90-day study in Japan. The first six datapoints are for the baseline measurements and the first 5 days in the study site, followed by three more measurements at the 30-day, 60-day, and 90-day check-ins. Red circles show data for participants who continued to smoke, green triangles data for the cessation group, and yellow squares show data for participants switching to THS.
For some of the HPHCs, the data from the 90-day studies indicated a slight increase in the levels of the relevant biomarkers at day 30 compared to day 5 for all groups. Considering that all study participants were current smokers at the time of enrollment in the study, their home environment may contribute to their exposure—which could include exposure from food, pollution, secondhand smoke, or other sources that were not present at the study site.
What’s next?
These studies demonstrate that the lower levels of harmful chemicals that are present in THS aerosol do indeed result in reduced exposure, as measured for the biomarkers reported in these studies. To provide additional evidence on this point, similar assessments are also being conducted on later versions of the THS.
Importantly, a reduction in exposure to HPHCs doesn’t automatically mean a reduction in harm. To gain an understanding of the potential positive impact on smokers’ health as a result of switching from cigarettes to THS, we have conducted a 1-year exposure response study that measured eight biomarkers of potential harm. In this study, all eight biomarkers of potential harm showed favorable changes in the same direction as with smoking cessation. Learn more about the effects of switching to THS on biomarkers of potential harm in that study.